Again, this is part of clinical governance but deserves its own page
Serious incidents may be defined as:
Acts or omissions in care that result in
- unexpected or avoidable death
- unexpected or avoidable injury —> serious harm
- Abuse
- Never events
- Incidents which prevent or treaten to prevent an organisations ability to continue to deliver an acceptable quality of healthcare services and incidents that cause widespread public concern results in a loss of confidence in services
Action points
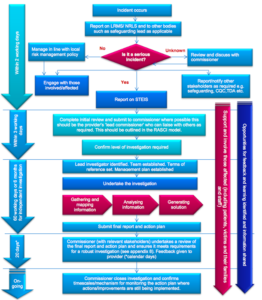
- Declare internally
- Take immediate action to establish facts
- Ensure safety of patients and staff
- Secure relevant evidence to support further investigation
- Disclose as soon as possible to patient, family or carers
- Inform the commissioner within 2 working days
- Undertake a RCA – there are 3 levels
1) concise investigations
2) multidisciplinary team investigations
3) independant investigation
Should be completed within 60 days and independent Ix within 6 months
Framework: 3 parts
1) Definitions and Thresholds
- NB no definitive list although there is a list of NEVER events:
- wrong site surgery
- wrong implant/prosthesis
- Retained FB post procedure
- Mis-selection of a strong potassium containing solution
- Wrong route administration of medication
- Overdose of insulin due to abbreviations or incorrect device
- Overdose of methotrexate for non cancer treatment
- Inappropriate administration of high strength midazolam
- Failure to install functional collapsible shower or curtain rails in MH settings
- Falls from poorly restricted windows
- Chest or neck entrapment in bedrails
- Transfusion or transplantation of ABO incompatible blood components
- Misplaced naso or oro-gastric tubes
- Scalds
If unclear, should be frank and honest discussions
Near – miss – can be a serious incident – look at likelihood of incident happening again, also potential for harm
2) Underpinning principles
7 key principles
- Open and transparent
- “Being Open’ guidance
- Duty of Candour :Requires a culture of candour
- training and support for staff to disclose information about unanticipated events in a patient’s care and apologise when appropriate;
- improvements in the levels and accuracy of the reporting of patient safety incidents;
- spreading and applying lessons learned into practice and publicly reporting these.
- Preventative
- Objective
- Timely and responsive
- within 2 working days
- Systems based
- the what
- the how
- the why (root cause)
-
- Proportionate
- Collaborative
3) Serious incident Management process
There must be clear procedures for:
- Timely reporting and liaison with their commissioning bodies (incidents must be recorded on STEIS within 2 working days of being identified). Particular types of incidents may require additional reporting to other systems. See appendix 2.
- Compliance with reporting and liaison requirements with regulators and other agencies/partners. See appendix 2
- Mechanisms to support robust serious incident investigations, including processes to ensure the following:
- Early, meaningful and sensitive engagement with affected patients and/or their families/carers, from the point at which a serious incident is identified, throughout the investigation, report formulation and subsequent action planning through to closure of the investigation process. A specific person should be assigned to engage with the family to provide a single point of contact.
- Clear procedures for taking immediate action following a serious incident including the collection and retention of evidence i.e. notes/clinical records,
31 Quality in the New Health System, Maintaining and Improving Quality. National Quality Board, January 2013 26
OFFICIAL
written accounts/statements32 from those involved, equipment involved, information from the location (site visit) and interviews with relevant individuals.
o Investigations are undertaken by appropriately trained and resourced staff and/or investigation teams that are sufficiently removed from the incident to be able to provide an objective view.
o Investigations follow a systems-based approach to ensure any issues/problems with care delivery are fully understood from a human and systems factors perspective and that the ‘root causes’ are identified (where it is possible to do so) in order to produce focused recommendations that result in SMART (specific, measurable, attainable, relevant, time-bound) actions and learning to prevent recurrence.
o Access to relevant specialists/ experts, communications expertise, administrative support and/or additional resources to support investigations where required.
o Mechanisms to ensure that actions from action plans are monitored until implemented and there is evidence of whether or not the action plan has resulted in the practice / system improvement anticipated. This should include oversight of implementation by organisation leaders.
o Mechanisms to support investigations being led by external agencies such as the police, HSE or local authority. Where required, providers must submit evidence to contribute towards external investigations.
o Processes (including interagency investigation policy and/or memorandum of understanding with relevant organisations) to support collaboration and partnership working where joint investigations are required to avoid duplication of activity or confusion of responsibility.
o Quality assurance processes to ensure completion of high quality investigation reports and action plans to enable timely learning and closure of investigations and to prevent recurrence.
o Mechanisms and effective communication channels to facilitate the sharing of lessons learned across the organisation and more widely where required.
I realise this is a bit waffly, I will be coming back to this to update and clarify
Reference
http://www.england.nhs.uk/wp-content/uploads/2015/04/serious-incidnt-framwrk-upd.pdf